The UK, Canada and the United States have accused Russia of spying on their Covid-19 vaccine research works through cyber attacks.
There were repeated cyber attacks by a Russian hacking group linked to the Russian government on academic and pharmaceutical research institutions involved in the coronavirus vaccine research, intelligence reports said.
The hacking group called APT29, also known as Cozy Bear and said to be part of the Russian intelligence service, is said to be responsible for series of cyber attack on institutions involved in coronavirus vaccine development.
Britain’s National Cyber Security Centre made the announcement, which was coordinated with authorities in the US and Canada.
“It is completely unacceptable that the Russian Intelligence Services are targeting those working to combat the coronavirus pandemic,” Foreign Secretary Dominic Raab said in a statement. “While others pursue their selfish interests with reckless behaviour, the UK and its allies are getting on with the hard work of finding a vaccine and protecting global health.”
Russia Denies Allegations
Moscow rejected the allegations, saying the claims were fabricated.
“We have no information on who could have hacked pharmaceutical companies and research centres in Britain,” Kremlin spokesman Dmitry Peskov told the TASS news agency.
“We can only say this: Russia has nothing to do with these attempts.”
It is not clear whether any information was stolen but the British intelligence says individuals’ confidential information is not believed to have been compromised.
Cozy Bear Hacker Group

Cozy Bear is a Russian hacker group classified as advanced persistent threat APT29, believed to be associated with Russian intelligence.
The Dutch AIVD deduced from security camera footage that it is led by the Russian Foreign Intelligence Service (SVR).
Cybersecurity firm CrowdStrike also previously suggested that it may be associated with either the Russian Federal Security Service (FSB) or SVR.
The group was given other nicknames by other several cybersecurity firms.
Russian Defense Ministry’s clinical trials of COVID-19 vaccine enter final stage
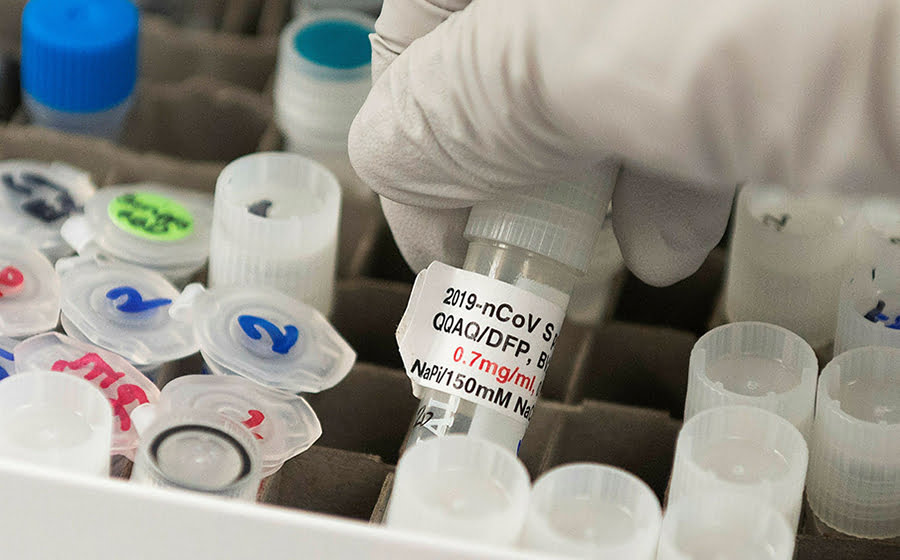
The Russian Defense Ministry and the Gamalei National Research Center for Epidemiology and Microbiology have started the final stage of clinical trials of a vaccine against the novel coronavirus, the ministry stated on Friday.
According to the Defense Ministry, “an in-ward treatment of the first group of volunteers, who were tested for the safety and tolerability of the vaccine, will end on July 15.”
“On Monday, July 13, the second group of volunteers, who are tested for the efficiency and immunogenicity of the vaccine, will be injected with the second component of the vaccine against the coronavirus,” the ministry stated.
“The booster scheme of the vaccination, which is intended for the second group of volunteers, will enable to strengthen the immune system and will also prolong its endurance,” according to the ministry.
The ministry also added that all volunteers feel well, have no complaints; they experience no side-effects at all and the first group of volunteers would be discharged from the Burdenko Military Hospital after undergoing the final tests.
Results of the COVID-19 vaccine tests, performed on a group of volunteers in Russia, show that they are developing immunity to the coronavirus, the ministry continued.
“In line with the research protocol, volunteers regularly take tests for antibody and cell-mediated immunity,” the ministry stated. “The data obtained by the Gamalei National Research Center for Epidemiology and Microbiology, proves that volunteers of the first and second groups are forming an immune response after injections of the vaccine against the coronavirus.”
The Defense Ministry announced in early June that two groups of volunteers had been selected for the clinical tests of an anti-coronavirus vaccine. The first group of 50 servicemen includes five women and ten medics. The second group is comprised of civilians.
Clinical testing of the vaccine developed by the Gamalei National Research Center for Epidemiology and Microbiology began on June 18. Eighteen volunteers were vaccinated against the novel coronavirus.







![[Full Transcript] Trump Pressures Raffensperger to Find 11,779 Votes to Overturn Biden's Win in Georgia 10 [Full Transcript] Trump Demands Raffensperger to Find 11,779 Votes to Overturn Biden's Win in Georgia US Stops Funding WHO Over Allegations of China Covid-19 Cover up Coronavirus: US overtakes Italy as country with most deaths](https://reportafrique.com/wp-content/uploads/2020/04/trump-260x195.webp)


